What is Humulin?
Humulin is synthetic human insulin prepared by using genetic engineering. Humulin is manufactured from DNA sources in laboratory, using recombinant DNA technology. Synthetic insulin is also called genetically engineered insulin. The synthetic insulin (humulin) is as effective as hormone insulin secreted by human pancreas.
Synthesis of Humulin
In 1978, scientists synthesized human insulin from E.coli bacteria using recombinant DNA technology, by preparing two DNA sequences for A and B chains of human insulin and introduced them in plasmid of E.coli. This led to production of human insulin chain. Eli Lilly, an American company marketed the first human insulin called humulin in 1983. Eli Lilly and Ranbaxy launched a new insulin project namely Humalog (an analog of 5, 6 human insulin), which is more expensive than human insulin products, but have good absorption in body, as compared to other insulin products.
Genetically engineered Insulin : Humulin, preferred over old animal based products
Structure of Insulin
Insulin is a proteinaceous hormone secreted by beta-cells of islets of langerhans of pancreas. Insulin controls, blood sugar level and when there is less secretion of insulin, it results in diabetes (high blood- sugar level).
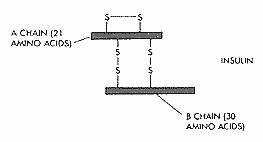
In 1954, Frederick Sanger determined primary structure of Insulin. Insulin is a protein formed by two polypeptide chains: A-chain and B-chain, interlinked by two sulphide bonds (see fig). A-chain is formed of 21 amino acid residue, while B – chain is formed of 30 amino acid residue. The A-chain has N-terminal glycine (GLY) and a C-terminal Asparagine (Asn), while the B-chain has an N-terminal phenylalanine (Phe) and a C terminal Alanine (Ala). Two disulphide bonds (-S-S-) present between two chains lie between cysteine amino acids located at 7th and 20th position of A-chain and 7th and 19th position of B-chain. A third disulfide bond also occurs in the A-chain between cysteine (Cys) amino acids at 6th and 11th position.
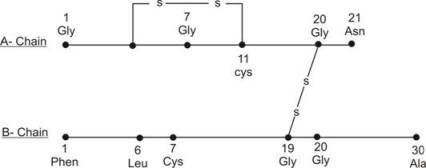
Primary structure of human insulin
Both chains of insulin are biosynthesized as a single polypeptide chain called proinsulin (inactive insulin), in which A and B chains are interlinked by a connecting polypeptide of 33 amino acids.
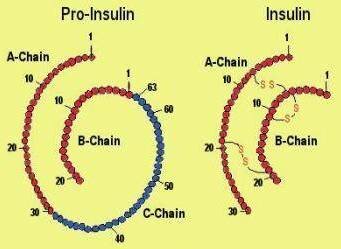
Proinsulin where A-chain and B-chain connected by C-chain and insulin without C-chain
Credit : Link
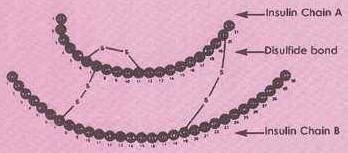
Active Insulin, Chance, R. and Frank B. – Research, development, production and safety of Biosynthetic Human Insulin.
Human insulin molecule. Source: Watson, J.D., Gilman, M., Witkovski, J., Zoller, M. – Recombinant DNA, pg 456.
Synthesis of Insulin is controlled by the gene located on the short arm of chromosome 11. It then undergoes proteolytic processing forming Insulin wherein deletion of connecting polypeptide from proinsulin takes place.
Advantages of Humulin over Insulin
Earlier insulin required for diabetes was extracted from pancreas of slaughtered cattle, pigs or salmon. The process was quite tedious and difficult and yields of insulin would be low. This extracted insulin in some patients, developed allergy or other side effects due to foreign protein. Due to disadvantages of animal insulin and advantages of humulin, humulin is regarded superior to animal insulin Humulin is considered better than animal insulin because:
- Humulin is absorbed more rapidly and show its effectiveness in short duration.
- Humulin causes fewer allergic and autoimmune reactions as compared to animal insulin.
- Humulin is less expensive than animal insulin
It is due to above advantages, now almost all insulin marketed is human insulin.
